Req 2a — Soap & Alcohol
When you get a cut or scrape, one of the first things someone tells you is “wash it with soap and water” or “put some rubbing alcohol on it.” But have you ever stopped to think about why those two substances are used? The answer comes down to chemistry.
Why Soap?
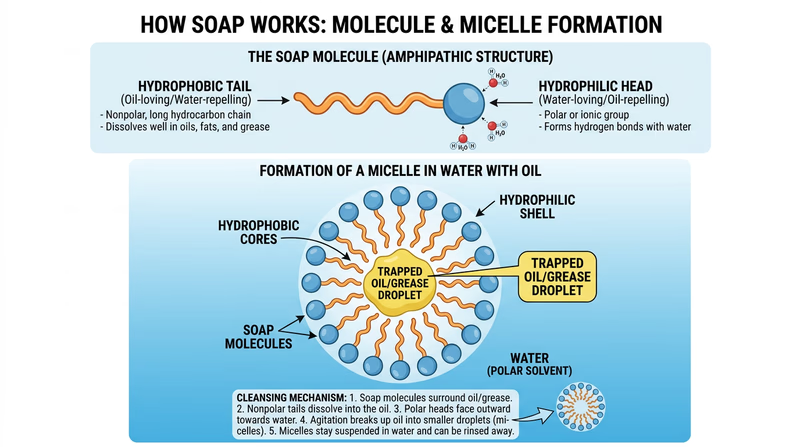
Soap is a surfactant — a molecule that has two ends with very different properties. One end is hydrophilic (water-loving) and the other is hydrophobic (water-fearing, but oil-loving). This dual nature is what makes soap so effective.
When you wash a wound with soap and water:
- The hydrophobic tails of soap molecules attach to dirt, oil, and bacteria on your skin.
- The hydrophilic heads stay dissolved in the water.
- As the water rinses away, the soap molecules carry the dirt and bacteria with them.
Soap does not technically “kill” most bacteria — it removes them from your skin mechanically. Think of it like using a mop instead of a flamethrower. The bacteria get lifted off your skin and washed down the drain.
Why Rubbing Alcohol?
Isopropyl alcohol (rubbing alcohol) works differently from soap. Instead of removing bacteria, it kills them by destroying their cell membranes. When alcohol contacts a bacterial cell, it dissolves the fatty outer membrane, causing the cell to fall apart and die.
Alcohol is also effective against many viruses for the same reason — it breaks apart the lipid (fat) envelope that surrounds many viral particles.
However, alcohol has some limitations:
- It can sting and irritate damaged skin tissue
- It evaporates quickly, so its germ-killing action is brief
- It is less effective against certain types of bacteria that have tough, protective outer coats (called spores)
Forming a Hypothesis
This requirement asks you to think about why soap and alcohol are used and then discuss how you might test your ideas. In science, a testable idea is called a hypothesis. Here is how to think about it:
- Observe — Soap and alcohol are both used to clean wounds, but they work differently.
- Ask a question — “Does soap remove more germs than alcohol kills?”
- Form a hypothesis — “I think soap removes more bacteria because it physically lifts them away, while alcohol only kills bacteria it directly contacts.”
- Design a test — You could swab a surface, apply soap to one area and alcohol to another, and then culture the swabs to see which has fewer bacteria remaining.
The Scientific Method in Action
This requirement is really about using the scientific method, which is the foundation of all chemistry:
- Observation — Notice something interesting
- Question — Ask why or how
- Hypothesis — Propose an explanation
- Experiment — Test your hypothesis
- Analysis — Look at the results
- Conclusion — Did the data support your hypothesis?
You will get to actually run experiments in the next two requirements, but forming the hypothesis first is where real science begins.