Req 2b — How Soap Works
This is one of the most satisfying experiments in the Chemistry merit badge. You are going to see — right on your own hands — why soap is so much better at cleaning than water alone. The answer lies in the chemistry of polarity.
Why Water Alone Falls Short
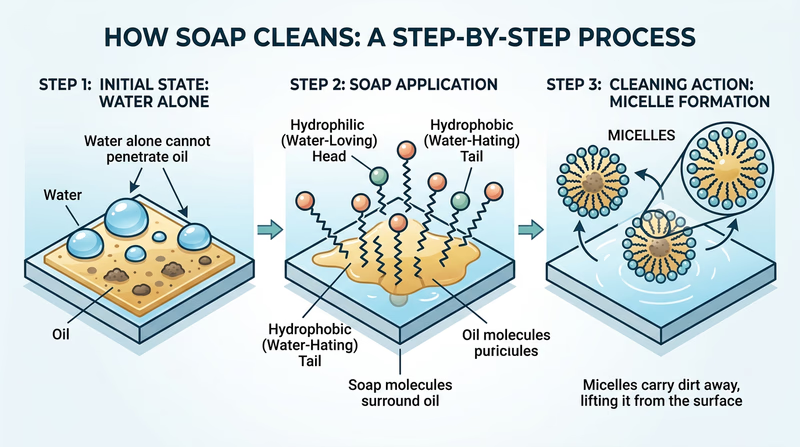
Water is sometimes called the “universal solvent,” but it has a major weakness: it cannot dissolve oil. Water molecules are polar, meaning they have a positive end and a negative end. Oil molecules are nonpolar — they have no charged ends. Polar and nonpolar molecules do not mix. That is why oil and water separate in a glass, and it is why water alone slides right off your oily hands.
When you coat your hands with cooking oil and powder, then rinse with just water, you will notice:
- The water beads up and rolls off the oily surface
- The powder stays stuck in the oil layer
- Your hands still feel greasy after rinsing
How Soap Bridges the Gap
Soap molecules are special because each one has both a polar end and a nonpolar end. This structure lets soap act as a bridge between oil and water:
- The nonpolar tail of the soap molecule digs into the oil on your skin.
- Many soap molecules surround a tiny droplet of oil, with their tails pointing inward and their polar heads pointing outward toward the water.
- This creates a tiny sphere called a micelle — a ball of soap molecules with oil trapped inside.
- Because the outside of the micelle is polar (water-friendly), the whole thing dissolves in water and rinses away, taking the oil and dirt with it.
When you coat your hands again and wash with soap and water, you will notice:
- The soap lathers up and mixes with the oil
- The powder lifts off your skin with the oil
- Your hands feel completely clean after rinsing
Running the Experiment
Experiment Materials
What you will need
- Cooking oil: Vegetable oil, olive oil, or coconut oil all work well.
- Nontoxic powder: Cocoa powder, cinnamon, sand, or cornstarch. Cocoa powder is easiest to see.
- Liquid hand soap: Regular hand soap or dish soap.
- Paper towels: For cleanup.
- A sink: With warm running water.
What to Observe and Discuss
When you talk with your counselor, be ready to describe:
- What happened with water only — Did the oil come off? Did the powder stick? How did your hands feel?
- What happened with soap and water — How quickly did the oil come off? Was the powder removed? How did your hands feel afterward?
- Why the difference? — Explain the concept of polarity and micelles in your own words.
- Real-world connection — How does this relate to cleaning a wound? Oil, dirt, and bacteria on your skin behave just like the cooking oil and powder in this experiment.