Req 3a — The Maillard Reaction
Every time you toast bread, sear a steak, or bake cookies, you are performing one of the most important chemical reactions in cooking: the Maillard reaction. Named after French chemist Louis-Camille Maillard, who described it in 1912, this reaction is responsible for the golden-brown color and incredible flavors you associate with perfectly cooked food.
What Is the Maillard Reaction?
The Maillard reaction occurs when amino acids (building blocks of proteins) react with reducing sugars (like glucose or fructose) at high temperatures — typically above 280°F (140°C). This reaction produces hundreds of new compounds that give browned food its distinctive color, aroma, and flavor.
It is not just one reaction — it is a cascade of many reactions happening simultaneously, creating an enormous variety of flavor molecules. That is why a grilled burger tastes completely different from raw ground beef, even though it is made of the same ingredients.
Physical Change vs. Chemical Change
This is the core chemistry concept for this requirement. Understanding the difference between physical and chemical changes is fundamental to all of chemistry.
Physical change: The substance changes its appearance or form, but its molecular structure stays the same. You can usually reverse a physical change. Examples: melting ice (water is still H₂O), dissolving sugar in water, cutting paper.
Chemical change: The substance’s molecular structure is permanently altered, creating entirely new substances. Chemical changes are usually irreversible. Signs of a chemical change include color change, new odor, gas production, heat release, and formation of a precipitate.
The Maillard reaction is a chemical change because:
- New molecules are created (flavor compounds, pigments)
- The color changes permanently (you cannot un-toast bread)
- New aromas are produced
- The nutritional composition changes
- Heat energy drives the transformation
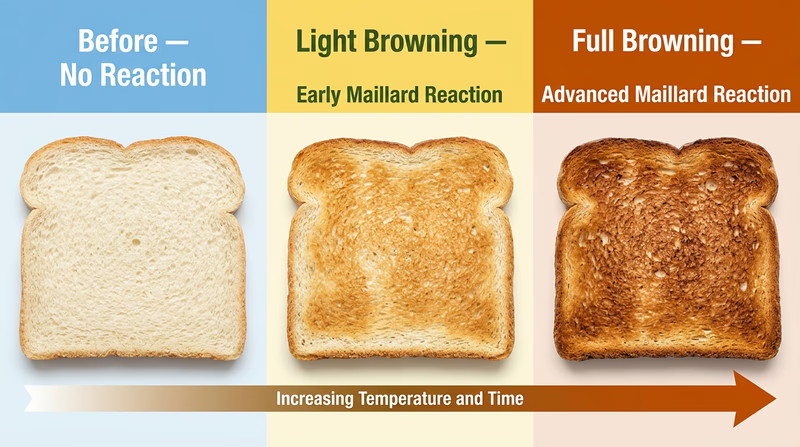
Performing the Reaction
The simplest way to perform a Maillard reaction is to toast a piece of bread. But you can also try any of these:
- Pan-sear a piece of meat or mushroom — Watch the surface transform from pale to deep brown.
- Caramelize onions — Slowly cook sliced onions in a pan until they turn golden-brown and sweet.
- Bake cookies — Notice how the dough changes from pale to golden-brown at the edges.
- Roast marshmallows — A camping favorite that perfectly demonstrates browning.
What Happens at the Molecular Level
Here is what is happening inside your food during the Maillard reaction:
- Heat breaks bonds — The thermal energy breaks apart amino acids and sugars into reactive fragments.
- New bonds form — These fragments recombine in new ways, creating molecules that did not exist before: melanoidins (brown pigments), pyrazines (nutty, roasted flavors), furanones (caramel flavors), and many more.
- The reaction cascades — Each new compound can react further, creating even more compounds. This is why longer cooking produces deeper, more complex flavors.
- Carbon dioxide and water are released — These gases escape as steam, which is why bread loses weight when toasted.
Maillard Reaction vs. Caramelization
These two browning reactions are often confused, but they are chemically different:
| Feature | Maillard Reaction | Caramelization |
|---|---|---|
| Requires | Amino acids + sugars | Sugars only (no protein needed) |
| Temperature | Above 280°F (140°C) | Above 320°F (160°C) |
| Examples | Seared steak, toasted bread, roasted coffee | Crème brûlée, caramel sauce, cotton candy |
| Flavors | Savory, nutty, roasted, meaty | Sweet, buttery, toffee-like |
Both are chemical changes, but the Maillard reaction requires protein (amino acids), while caramelization only involves sugar.