
Nuclear Science Merit Badge — Complete Digital Resource Guide
https://merit-badge.university/merit-badges/nuclear-science/guide/
Introduction & Overview
Overview
Nuclear science is the study of matter, energy, and tiny particles that are too small to see but powerful enough to light cities, diagnose disease, and help scientists understand how the universe works. This badge asks you to look past movie myths and learn what radiation really is, how atoms behave, and why careful measurements matter. If you like science that connects the invisible world to real life, Nuclear Science is full of moments that make you stop and say, “Wait, really?”
Then and Now
Then
People have always lived in a radioactive world. Long before anyone knew what an atom was, the sun, rocks, soil, and even the human body were quietly producing or interacting with radiation. The big change came in the late 1800s, when scientists such as Henri Becquerel and Marie Curie realized that certain materials gave off energy on their own. Soon after, Ernest Rutherford and other researchers began uncovering the structure of the atom and showing that matter was not solid and simple after all.
Early nuclear science felt mysterious because the tools were simple and the effects were invisible. Researchers used photographic plates, gold leaves, glowing screens, and careful counting to detect particles they could not see directly. Those classic experiments are still important because they show how scientists learned to trust evidence over guesses.
Now
Today, nuclear science touches daily life in ways many people never notice. Doctors use radioactive tracers to look inside the body. Engineers use radiation to inspect welds and materials without cutting them open. Scientists use particle accelerators to study the building blocks of matter. Nuclear reactors produce electricity without burning fuel the way coal or gas plants do.
Modern nuclear science is also built on safety culture. The field depends on measurement, shielding, planning, and clear procedures. That is why this badge spends so much time on radiation basics and dose control before moving into experiments and energy.
Get Ready!
This badge rewards curiosity and careful thinking. You do not need to memorize every term on day one, but you do need to get comfortable asking good questions, drawing what you cannot see, and backing up your ideas with observations. By the end, you should feel more confident talking about radiation without fear or exaggeration.
Kinds of Nuclear Science
Radiation and Health
One part of nuclear science is learning what radiation is, where it comes from, and how it affects people and living things. This includes background radiation, contamination, dose, and medical uses such as imaging and treatment.
Atomic Structure and Isotopes
Another part of the badge is about the structure of matter itself: protons, neutrons, electrons, quarks, and isotopes. These ideas help explain why some atoms are stable while others decay and release radiation.
Particle Physics and Research Tools
Particle accelerators let scientists push particles to high energies and study what happens when they collide or pass through targets. This branch of nuclear science asks some of the biggest questions in physics: What is matter made of? How do forces work? What can we learn from nuclei and nucleons?
Energy and Useful Applications
Nuclear science is not only about reactors. It includes medicine, industry, food science, environmental monitoring, and space exploration. One reason this badge is interesting is that the same basic ideas can help both a doctor in a hospital and a scientist studying cosmic rays.
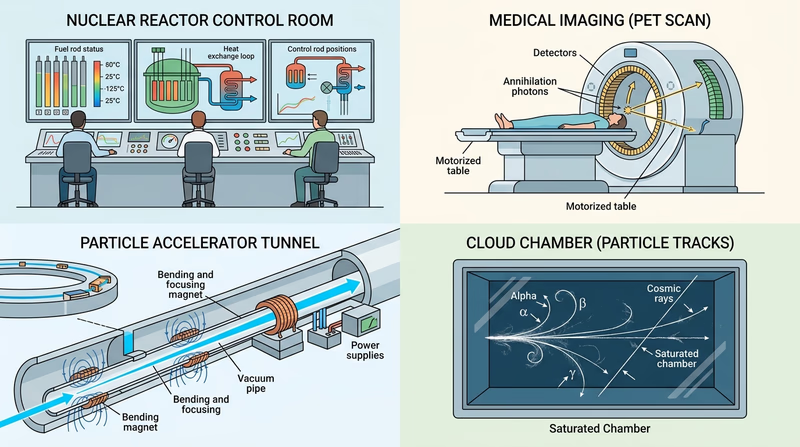
Ready to start with the most important foundation in the whole badge? Begin with what radiation is, where you meet it in everyday life, and how to think about it clearly.
Req 1 — Understanding Radiation
This requirement covers the ideas that make the rest of the badge possible:
- What radiation is and how it differs from ordinary light, heat, and radio waves
- How to work safely by keeping dose as low as reasonably achievable
- What warning symbols mean and where they belong
- Where radiation comes from in daily life
- How exposure, contamination, and dose differ
If you can explain these five parts clearly, you will sound much more like a scientist and much less like someone repeating movie myths.
Requirement 1a
Radiation is energy moving from one place to another. Sometimes that energy travels as waves, and sometimes it travels as particles. Sunlight is radiation. Heat from a campfire is radiation. So are alpha particles and gamma rays.
The key difference in this badge is whether the radiation has enough energy to knock electrons off atoms. If it does, it is ionizing radiation. If it does not, it is nonionizing radiation.
| Type | Examples | What it can do |
|---|---|---|
| Nonionizing | radio waves, microwaves, visible light, most ultraviolet | move energy around, heat things, let you see |
| Ionizing | alpha particles, beta particles, gamma rays, X-rays | remove electrons from atoms and damage cells or materials |
A good way to remember it is this: nonionizing radiation can still matter, but ionizing radiation can change atoms directly. That is why nuclear science treats ionizing radiation with extra care.
🎬 Video: Radiation & You (video) — https://youtu.be/FrVUJ2c_Rwc?si=3e61M3Jbgg9w3ToV
🎬 Video: IDTIMWYTIM: Radiation (video) — https://youtu.be/uJ3ea9fa6CA?si=lpSg7dn3koa5wAOV
Requirement 1b
ALARA means As Low As Reasonably Achievable. It does not mean “be fearless” and it does not mean “panic about every tiny dose.” It means planning your activity so you receive as little radiation as practical while still doing the job.
The three main ALARA tools are simple:
Your ALARA game plan
Use this before any activity involving a source or detector- Time: spend only the amount of time needed near the source.
- Distance: stay as far away as you can while still making the observation or measurement.
- Shielding: put the right material between you and the source when the setup allows it.
- Procedure: know what you are going to do before you start, so you do not waste time figuring it out while exposed.
- Supervision: work only with trained adults and approved equipment.
A strong counselor answer sounds like a plan: “First I would understand the setup. Then I would gather materials, decide where everyone stands, make measurements efficiently, and step back when I am done.”
The ALARA Principle (website) A clear overview of why time, distance, and shielding work together to reduce dose. Link: The ALARA Principle (website) — https://www.cdc.gov/radiation-health/safety/alara.htmlRequirement 1c
The standard radiation hazard symbol is a trefoil: a center circle with three blades around it. It warns that ionizing radiation may be present and that special controls are needed. The exact colors may vary by setting, but the message is the same: stop, think, and follow trained procedures.
You should expect this symbol on places or items such as:
- doors to X-ray or radioactive materials rooms
- storage containers for licensed radioactive sources
- equipment that generates X-rays or uses radioactive material
- labels, transport containers, and controlled areas where regulations require it
The symbol should not be used as decoration or on things that are not real hazards. Warning labels only work if people trust them.
🎬 Video: Radiation Hazard Symbol (video) — https://youtu.be/Nss1nl56fPo?si=gjiyC4gVAGL9CMsk
Requirement 1d
You live in a radiation environment all the time. Some of it comes from space, especially cosmic rays striking Earth from outside the planet. Some comes from Earth itself, because rocks, soil, air, water, and living things contain naturally occurring radioactive atoms.
Examples of everyday exposure include:
- Cosmic radiation from space, especially at high altitude
- Radon gas that seeps from soil into buildings
- Natural radioactivity in rocks and soil
- Tiny amounts of radioactive isotopes in food and in your body
Four good NORM examples a Scout might actually find are bananas, potatoes, Brazil nuts, and granite countertops. These are radioactive because they contain trace amounts of naturally occurring isotopes such as potassium-40, uranium, thorium, or their decay products. The word “radioactive” sounds dramatic, but in everyday materials the amounts are usually very small.
🎬 Video: Five Radioactive Products We Use Every Day (video) — https://youtu.be/-9gD6FMivTQ?si=O-BCuqRKLgTMGZpg
🎬 Video: Your Grocery Store is Radioactive (and So Is Everything Else) (video) — https://youtu.be/FZtR3UEEkXA?si=jC5BJJVPgtDS4nK3
🎬 Video: The Mystery of High-Energy Cosmic Rays (video) — https://youtu.be/_bKbMARsE-4?si=ba7MFiT_UCPs9WZj
Requirement 1e
Exposure means radiation is passing through or reaching you. Contamination means radioactive material is actually on you, in you, or where it should not be. A person can be exposed without being contaminated, just as you can stand in sunlight without carrying the sun away with you.
The hazards depend on the type of radiation, how much dose is received, how long the exposure lasts, and whether contamination gets inside the body. Large doses can damage cells and tissues. Contamination can spread through air, water, soil, or food webs if it is not contained. Wildlife can be affected for the same reasons people are: radiation can damage living tissue and change how organisms grow, reproduce, or survive.
For your dose comparison, use a trusted calculator and pay attention to units. A useful answer is not just a number. It is a comparison with context, such as whether your annual background dose is lower than, similar to, or higher than an occupational dose limit or a typical worker’s monitored dose.
🎬 Video: What Do Radiation Contamination and Exposure Mean? (video) — https://youtu.be/Ry2YpGjnakg?si=zaj2augqvJll6mwb
🎬 Video: Biological Effects of Radiation (video) — https://youtu.be/EuKzI3g5ra4?si=0z8dwKUGPc34S2YD
Now that you know what radiation is and how to think about it safely, the next step is to zoom in and study the atom itself.
Req 2 — Atoms, Isotopes, and Particles
This requirement gives you the vocabulary and models that make the rest of the badge easier to understand. First you learn the parts of matter and the language scientists use. Then you turn those ideas into something you can build and explain with your own hands.
Requirement 2a
A strong explanation connects the terms instead of listing them like flash cards.
An atom is the basic unit of an element. At its center is the nucleus, made of protons and neutrons. Electrons move around the nucleus in the surrounding electron cloud. Protons and neutrons are built from smaller particles called quarks.
An isotope is a version of the same element that has the same number of protons but a different number of neutrons. Some isotopes are stable. Some are unstable and change over time.
When an unstable nucleus changes and gives off energy or particles, that is radioactivity. A radioactive isotope is a radioisotope.
The main emissions you need to know are:
| Term | What it is | What to remember |
|---|---|---|
| Alpha particle | 2 protons + 2 neutrons | heavy, charged, stopped easily |
| Beta particle | fast electron or positron | lighter, travels farther than alpha |
| Gamma ray | high-energy electromagnetic radiation | no mass, very penetrating |
| X-ray | high-energy electromagnetic radiation usually produced outside the nucleus | also penetrating, often made by equipment |
Ionization happens when enough energy knocks electrons off atoms. Stability describes whether a nucleus tends to stay the same or decay.
Glossary of Nuclear Science Terms (website) A plain-language glossary you can use to check your definitions and make sure you are using each term correctly. Link: Glossary of Nuclear Science Terms (website) — https://nplab.webspace.durham.ac.uk/glossary/🎬 Video: What Are Isotopes? (video) — https://youtu.be/faB9Gb7bl9I?si=Et7Yi1cp8zLvyppP
🎬 Video: Quarks Explained (video) — https://youtu.be/LraNu_78sCw?si=o4zrlNUwhUT69eB6
Requirement 2b
This is where the badge stops being only words and turns into a model you can point to. Pick an element with several common isotopes so the pattern is easy to see. Hydrogen, carbon, oxygen, and chlorine all work well.
How to choose your element
Look for an element where the atomic number stays the same but the mass number can change. The atomic number tells you how many protons the element always has. The mass number is protons plus neutrons. Change the neutrons, and you have a new isotope of the same element.
What your three models should show
What to label on each isotope model
Make the differences easy to see- Protons: these identify the element.
- Neutrons: these are what make one isotope different from another.
- Electrons: for a neutral atom, these match the proton count.
- Atomic number: the proton count.
- Mass number: protons plus neutrons.
- Isotope notation: for example, carbon-12 or (^12_6\text{C}).
A good set of models does not need to be fancy. Colored clay, beads, paper circles, or a digital drawing can all work if the structure is clear.
Showing quarks
In your separate model or diagram, show that:
- a proton is made of two up quarks and one down quark
- a neutron is made of one up quark and two down quarks
You do not need to explain every detail of particle physics. The goal is to show that protons and neutrons are not indivisible. They are made from smaller parts.
Make a Model of an Isotope (PDF) A step-by-step activity sheet that can help you organize your isotope models and notation. Link: Make a Model of an Isotope (PDF) — https://filestore.scouting.org/filestore/Merit_Badge_ReqandRes/Requirement%20Resources/Nuclear%20Science/Isotope%20Model%20Activity.pdf Interactive Isotopes (website) Lets you explore real isotope data and compare proton, neutron, and mass-number patterns before you build your model. Link: Interactive Isotopes (website) — https://isotopes.ans.org/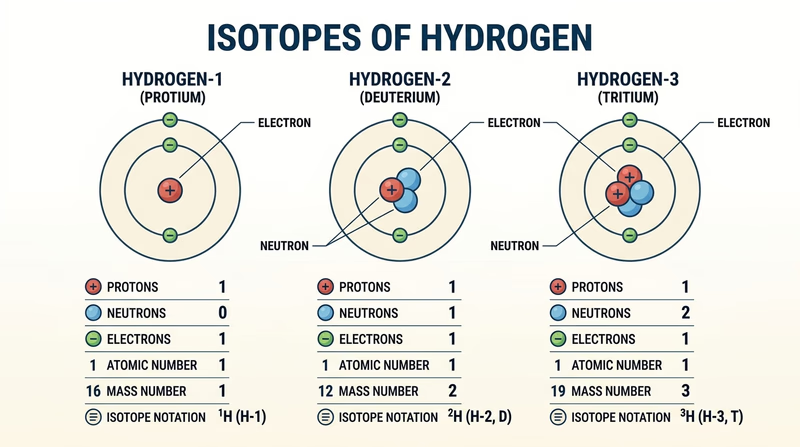
Once you are comfortable with atoms and isotopes, you are ready to look at machines that hurl particles at incredible speeds to learn even more.
Req 3a — How Accelerators Work
A particle accelerator is a machine that gives charged particles more and more energy, then guides them where scientists want them to go. Think of it like a super-precise racetrack for tiny particles. Instead of using gasoline, it uses electric fields to speed particles up and magnets to steer and focus them.
At a simple level, an accelerator does four jobs:
What an accelerator must do
Four jobs every particle accelerator handles- Create particles such as electrons, protons, or ions.
- Speed them up using electric fields.
- Steer and focus them using magnets so the beam stays on target.
- Deliver the beam to a target, a detector, or another beam for collision.
Why do scientists bother? Because fast-moving particles reveal information that slower ones cannot. When particles collide, scatter, or trigger detectors, scientists can learn about the structure of nuclei, the forces inside matter, and the properties of particles that are far too small to see directly.
There are different accelerator designs. Linear accelerators send particles down a straight path. Circular accelerators bend them around a loop so they can gain energy again and again. In both cases, the beam must be carefully controlled. If the beam spreads out, hits the wrong target, or loses too much energy, the experiment becomes harder to interpret.
What scientists study with them
In nuclear science, accelerators can:
- probe the inside of nuclei
- create rare isotopes
- study how nucleons behave
- test materials and detectors
- produce beams for medicine or industry
A good explanation for your counselor should connect the machine to the science: particles gain energy, magnets control the beam, detectors collect evidence, and scientists use that evidence to answer questions about matter.
🎬 Video: Particle Accelerators: What Are They, How Do They Work and Why Are They Important To Us? (video) — https://youtu.be/vIeRLeQq7V4?si=wf3TBAQxCGd1nYA5
Now you get to choose how you want to explore accelerator science: by talking with people who do the work or by comparing major machines and experiments.
Req 3b — Choose an Accelerator Path
For this requirement, you choose exactly one option. Both paths teach you how accelerator science is used in the real world, but they fit different kinds of Scouts.
Your Options
- Req 3b1 — Visit a Lab or University: Meet or speak with a scientist and learn how real researchers study nuclei or nucleons. This option is great if you like hearing directly from people doing the work.
- Req 3b2 — Compare Major Accelerators: Research three accelerators and the experiments they support. This option is great if you enjoy reading, comparing, and organizing information.
How to Choose
Choosing your option
Pick the path that matches your access and interests- Best for in-person learners: choose 3b1 if you can visit a university, national lab, or research facility, or arrange a conversation with a scientist.
- Best for independent researchers: choose 3b2 if travel is difficult but you enjoy comparing major facilities and their discoveries.
- What you will gain with 3b1: a first-hand look at how scientists talk about their work, solve problems, and use equipment.
- What you will gain with 3b2: a broader view of the field and a better sense of how different accelerators serve different purposes.
Before you choose, these two official videos can help you picture what large accelerator facilities look like and why scientists use them.
🎬 Video: Lawrence Berkeley National Laboratory Nuclear Science Division (video) — https://youtu.be/N3i6CgUHSoc?si=tXJzA1pfqwIYRlXE
🎬 Video: Inside the World's Largest Science Experiment (video) — https://youtu.be/nrXhK3Gh5EE?si=kH7N_aBRmhyZOkpJ
If you want the people-and-places version of this requirement, start with the visit option first.
Req 3b1 — Visit a Lab or University
This option is about learning how real scientists think. Even a short visit can teach you a lot if you arrive ready to listen, observe, and ask questions that go beyond “What do you do here?”
What to look for during your visit
You do not need to understand every machine or equation. Focus on the investigation itself.
Good things to notice
Watch for these during the visit or conversation- What question they are trying to answer: Are they studying the nucleus, a specific isotope, or how nucleons behave?
- What tools they use: accelerator, detector, target, computer model, shielding, or counting equipment.
- What evidence they collect: tracks, counts, energy measurements, images, or data plots.
- Why the work matters: basic science, medicine, energy, materials research, or national security.
Questions worth asking
A good visit usually depends on good questions. Try asking some of these in your own words:
- What are you trying to measure or discover?
- How does your equipment help you study nuclei or nucleons?
- What part of the process is hardest to control?
- How do you know when your data are trustworthy?
- What safety rules matter most in your lab?
- What surprised you when you first started doing this work?
Turning the visit into a clear explanation
Afterward, be ready to summarize the experience in four parts:
- Where you went or whom you spoke with
- What they study
- How they study nuclei or nucleons
- Why that work matters
That structure keeps your report from turning into a random list of cool details.
If travel is not possible, the research-based option gives you another way to explore the same field.
Req 3b2 — Compare Major Accelerators
This option is less about visiting one place and more about seeing the range of work particle accelerators can do. The best answers compare machines with different purposes instead of picking three versions of the same idea.
A smart way to choose your three accelerators
Try to choose one from each category:
- A giant research accelerator for fundamental physics
- A nuclear science facility focused on nuclei or isotopes
- A medical or applied accelerator used in hospitals or industry
That mix makes it easier to explain both basic science and practical applications.
What to include for each accelerator
Your comparison notes
Collect these details for each machine- Name and location
- What particles it accelerates
- What question or problem it helps study
- One or more experiments or uses
- What practical result came from the work
Here are example categories you could use in your own research:
| Accelerator type | Possible focus | Practical connection |
|---|---|---|
| Large collider | fundamental particles and forces | detector technology, computing, medical imaging tools |
| Nuclear research facility | isotope behavior and nuclear structure | medicine, reactor science, materials testing |
| Medical accelerator | beams for treatment or isotope production | cancer care, diagnostic imaging |
How to talk about experiments
When the requirement says to describe experiments, do not stop at the machine name. Explain what the scientists were trying to learn. For example, were they testing how particles scatter, producing rare isotopes, or creating beams for therapy? Then explain the practical side. Did the work improve detectors, help create medical isotopes, or lead to better understanding of matter?
A strong counselor discussion sounds comparative: “This machine studies basic physics questions, while this one is used for isotope production, and this one helps treat cancer patients.” That shows you understand the field, not just three names.
Classic experiments helped scientists discover radiation with very simple tools. Next, you will choose two of those hands-on investigations for yourself.
Req 4 — Pick Two Classic Experiments
You must choose exactly 2 options from this requirement.
Your Options
- Req 4a — Build an Electroscope: Build a simple detector that shows how electric charge behaves and how radiation can discharge it.
- Req 4b — See Tracks in a Cloud Chamber: Watch ionizing radiation leave visible trails in a supersaturated vapor.
- Req 4c — Model Half-Life: Use a hands-on experiment to show how radioactive decay follows a pattern even when individual atoms decay randomly.
How to Choose
Choosing your two experiments
Think about materials, time, and what you want to understand- Best low-cost build: 4a usually uses simple household materials and teaches electrostatics clearly.
- Best wow-factor demonstration: 4b lets you see radiation tracks, but it takes more setup and careful adult help.
- Best for graphing and math-minded Scouts: 4c turns decay into data you can count and discuss.
- What you will gain: 4a builds understanding of charge and detection, 4b helps you visualize invisible particles, and 4c helps you explain probability, decay, and half-life.
Start with the electroscope if you want the simplest classic detector first.
Req 4a — Build an Electroscope
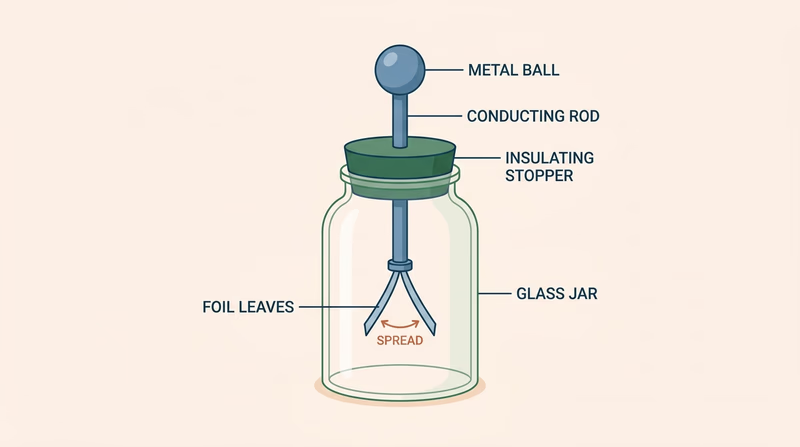
An electroscope is one of the simplest devices for showing that invisible things can produce measurable effects. Charge the metal part of the device, and the leaves or indicator separate because like charges repel. When ionizing radiation affects the air around the electroscope, the air becomes more conductive, and the stored charge leaks away faster.
What the electroscope is showing
The important idea is not just “the leaves move.” The important idea is why they move.
- A charged electroscope stores electric charge.
- The leaves spread apart because they carry similar charges.
- Ionizing radiation creates ions in the surrounding air.
- Those ions help charge move away, so the electroscope discharges.
That means the nearby radiation is not pushing the leaves directly. It is changing the air so the charge can escape.
🎬 Video: DIY Electroscope (video) — https://youtu.be/fCpaNzpFhPo?si=VwL6Mi6do8qlSrhI
What to explain when you demonstrate it
Focus on cause and effect- How you charged it
- What the leaves or indicator did at first
- What changed when the source moved nearby
- Why ionization makes the device lose charge faster
If you want a more dramatic visual of radiation, the next classic experiment lets you actually see tracks left behind in vapor.
Req 4b — See Tracks in a Cloud Chamber
A working cloud chamber feels like science fiction the first time you see it. Out of nowhere, thin white streaks appear and vanish in the chamber. Those streaks are evidence of ionizing particles moving through vapor and leaving a trail of ions behind them.
What is happening inside the chamber
A cloud chamber contains alcohol vapor cooled until it becomes supersaturated. That means the vapor is ready to condense but needs a trigger. When radiation passes through the chamber, it ionizes atoms along its path. The vapor condenses around those ions, making a tiny visible trail.
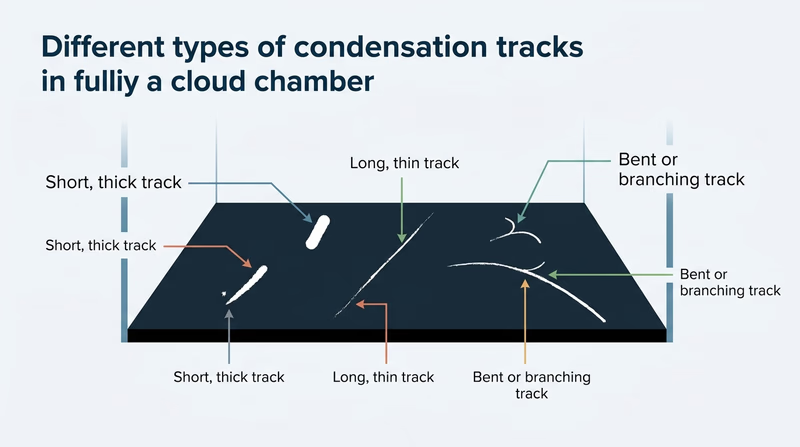
Different particles can leave different-looking tracks:
- short, thick tracks may suggest heavier particles
- longer, thinner tracks may suggest lighter or faster particles
- sudden bends or branching can hint at interactions along the way
You do not need to identify every track perfectly. What matters is explaining that radiation leaves behind ionization, and the vapor makes that invisible process visible.
🎬 Video: How to Build a Cloud Chamber! (video) — https://youtu.be/pewTySxfTQk?si=0aPyTx0YDHrocIAv
🎬 Video: Working Cloud Chamber | Exploratorium (video) — https://youtu.be/jh7SzrNWGhI?si=C4cCRtQruDkCGpAv
If you want to model radioactive decay with data instead of vapor trails, the next experiment is a great fit.
Req 4c — Model Half-Life
Half-life is one of the most important ideas in nuclear science because it shows how something can be random for each atom but still predictable for a large group. You cannot point to one unstable atom and know exactly when it will decay, but you can predict how a big sample will change over time.
What half-life means
A half-life is the time it takes for half of the radioactive atoms in a sample to decay. After one half-life, about half remain. After two half-lives, about one-fourth remain. After three, about one-eighth remain.
That pattern is why Scouts often model half-life with candies, coins, dice, or other objects. Each round stands in for a time interval. The exact items that “decay” each round change, but the general trend is clear.
🎬 Video: How Radioactive Decay Works | Decay Chains (video) — https://youtu.be/fo1tgibNLss?si=K2DrEfEaGW5MSdOR
🎬 Video: Skittles Half Life Lab | Radioactive Isotope Decay | Half Life Graph (video) — https://youtu.be/mCSXJNVdK0Y?si=A3UPqOjowKtkSmxk
How to discuss your experiment
Your counselor will likely want more than a graph. Be ready to explain the pattern.
What your discussion should include
Explain the idea, not just the procedure- What represented the atoms in your model
- What counted as decay during each round
- How the number changed over time
- Why the graph is not perfectly smooth but still shows the half-life pattern
What a decay chain is
A decay chain happens when one unstable isotope decays into another isotope that is also unstable. That new isotope may decay again, and the process can continue through several steps until a stable isotope is reached.
This matters because radiation hazards and measurements do not always come from just one isotope. Sometimes scientists must think about a whole family of related decay products.
If classic experiments showed how radiation can be detected and modeled, the next requirement shows how to protect people from it.
Req 5 — Choose a Safety Investigation
You must choose exactly one option from this requirement. Each one teaches radiation safety, but from a different angle.
Your Options
- Req 5a — Time, Distance, and Shielding: Use instruments and materials to see how dose rate changes with setup and protection.
- Req 5b — Radon at Home: Study a real household radiation issue that affects indoor air quality and long-term health.
- Req 5c — X-Ray Room Safety: Visit a place that uses X-rays and learn how room design protects workers and patients.
How to Choose
Choosing your safety investigation
Match the option to your access and interests- Best for hands-on measurement: 5a if your counselor has access to a survey meter and approved source.
- Best for home and community relevance: 5b if you want to study a hazard that many families can actually test for.
- Best for medical imaging interest: 5c if you can visit a clinic, dentist office, or hospital imaging area.
- What you will gain: 5a teaches direct control of dose, 5b teaches environmental health, and 5c teaches engineering controls and workplace safety.
Whichever option you choose, keep using the same safety mindset from Req 1: understand the setup, follow procedures, and think in terms of time, distance, and shielding.
Req 5a — Time, Distance, and Shielding
This requirement puts ALARA into action. Instead of talking about safety in theory, you measure how the setup changes the reading. That makes the three main protection tools feel real.
What counts per minute tells you
A survey meter detects radiation events and reports a reading such as counts per minute (CPM). A higher CPM usually means the detector is receiving more radiation events. If the source moves closer, the reading often rises. If it moves farther away, the reading often falls.
The reason distance matters is simple: radiation spreads out. When the detector is farther away, less of that radiation reaches it.
What shielding shows
Different materials reduce radiation differently because alpha, beta, and gamma radiation do not all interact with matter the same way. A light barrier may stop alpha radiation. Denser or thicker material may be needed for beta or gamma radiation.
When you compare three shielding materials, do not just list the readings. Explain why the differences happened. Was the material denser? Thicker? Better suited to that type of radiation?
How Materials Shield Radiation (PDF) A visual guide that helps you compare how different materials reduce radiation in different situations. Link: How Materials Shield Radiation (PDF) — https://filestore.scouting.org/filestore/Merit_Badge_ReqandRes/Requirement%20Resources/Nuclear%20Science/shutterstock_1169023357.pdf🎬 Video: Time, Distance and Shielding (video) — https://youtu.be/bNSKIbRNy3c?si=3WqSseNb_EbOeZAx
🎬 Video: Measuring Radiation (video) — https://youtu.be/GdZD_hJBdOE?si=p4mOulsJpBxDWH5j
🎬 Video: Using a Survey Meter: CPM vs Distance (video) — https://youtu.be/0-Yaeh3dOZk?si=Z1MokzEOznMdvlZD
Three ideas to say out loud
These are the real lesson of the activity- Time: less time near the source usually means less dose.
- Distance: more distance usually means a lower reading.
- Shielding: the right material can reduce what reaches the detector or person.
If you want to explore a radiation issue that shows up in ordinary buildings, the radon option is next.
Req 5b — Radon at Home
Radon is one of the most important everyday radiation topics because it can build up indoors without any smell, color, or warning sign. That makes testing essential. You cannot judge radon by guessing.
What radon is and why people test for it
Radon is a radioactive gas produced naturally as uranium in soil and rock breaks down. It can seep through foundations and collect inside basements and lower floors. The concern is long-term breathing exposure, especially over many years.
Short-term versus long-term tests
A short-term test is useful when you need a quicker snapshot, often over a few days. It can help screen a building or guide a next step. A long-term test runs much longer and gives a better picture of average conditions over time, because radon levels can change with weather, ventilation, and season.
| Test type | Best use | Main advantage | Main limitation |
|---|---|---|---|
| Short-term | quick screening or real-estate timeline | faster results | may not represent long-term average |
| Long-term | understanding normal exposure in a home | more accurate overall picture | takes longer |
How to talk through the testing steps
Your counselor will want to hear that you understand the process, not just the name of the test.
Steps to mention
A clear answer should include these parts- Where the test is placed: usually in a livable low level of the building, following kit directions.
- How long it stays in place: depends on whether it is short-term or long-term.
- Why conditions matter: doors, windows, HVAC use, and season can affect results.
- How results are read and interpreted: use the reporting instructions that come with the approved test method.
- What happens next: elevated results may lead to follow-up testing or mitigation.
Health concerns and mitigation
Breathing radon over time increases the risk of lung damage and lung cancer. The risk is especially serious for smokers, but radon is still a concern for nonsmokers too.
If radon levels are high, mitigation may include improving ventilation, sealing entry points, and installing a radon reduction system that vents gas safely out of the building.
🎬 Video: What is Radon, Anyway? (video) — https://youtu.be/24aoot_CwoM?si=IYOyLpbVk2z2fb4R
🎬 Video: How Dangerous Is Radon to Your Health? (video) — https://youtu.be/SlRVdKdTBFE?si=pJrBWumWDON3uPcG
If you want to see how radiation safety is designed into a workplace instead of a home, the X-ray room option is next.
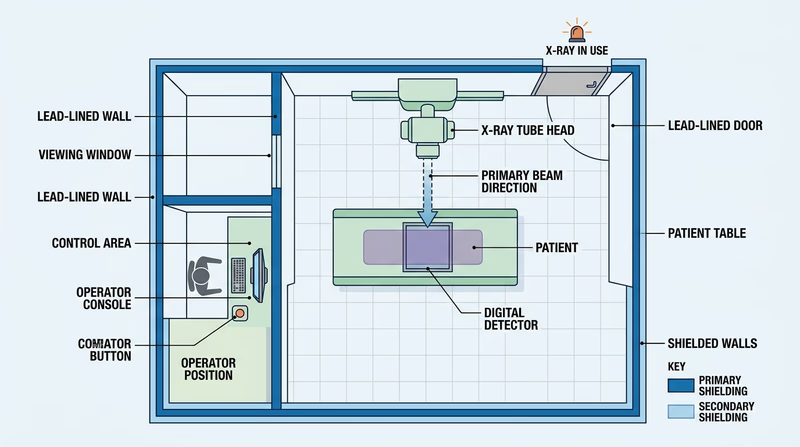
Req 5c — X-Ray Room Safety
This requirement shows that radiation safety is often built into the room before the machine is ever turned on. The layout, barriers, procedures, and operator position all work together.
What your floor plan should show
Your drawing does not need to be artistic. It needs to communicate where people and equipment are during an exposure.
Include these on your floor plan
Make the safety features obvious- The X-ray unit
- The patient location
- Where the operator stands during operation
- Protective barriers or control booth
- Doors, windows, and warning signs if they are part of the room layout
Common safety precautions
In many X-ray rooms, the operator stands behind a protective barrier or outside the room during exposure. The room may use lead-lined walls, lead glass, controlled distances, collimation to limit the beam, and shielding devices when appropriate.
These precautions matter because the operator may work around X-ray equipment every day. Even if each exposure is small, repeated unnecessary exposure is not acceptable. That is another real-world example of ALARA from Req 1b.
🎬 Video: X-Ray Room Tour || Ask the Rad Tech (video) — https://youtu.be/-pwY3-7lJJg?si=qctALb5U-IM9vG-K
You have now explored radiation protection from three different angles. Next, you will choose a path into nuclear energy.
Req 6 — Choose a Nuclear Energy Path
You must choose exactly one option. Both choices lead to the same big idea: nuclear energy becomes electricity by releasing heat, making steam, and turning turbines. The difference is whether you want to focus first on the physics of fission or the operation of a real plant.
Your Options
- Req 6a — Fission and Chain Reactions: Learn the physics behind splitting heavy atoms, critical mass, and reactor control.
- Req 6b — How Plants Make Electricity: Visit or research a plant and study how reactors fit into the larger power grid.
How to Choose
Choosing your nuclear energy option
Both options teach the same overall system from different angles- Best for model-builders and physics-minded Scouts: choose 6a if you want to understand the chain reaction itself.
- Best for real-world systems thinkers: choose 6b if you want to see how a plant, reactor, and electrical grid fit together.
- What you will gain with 6a: a clear explanation of fission, critical mass, and reactor control.
- What you will gain with 6b: a stronger grasp of plant operations, electricity generation, and energy comparisons.
Start with fission if you want to understand the physics first.
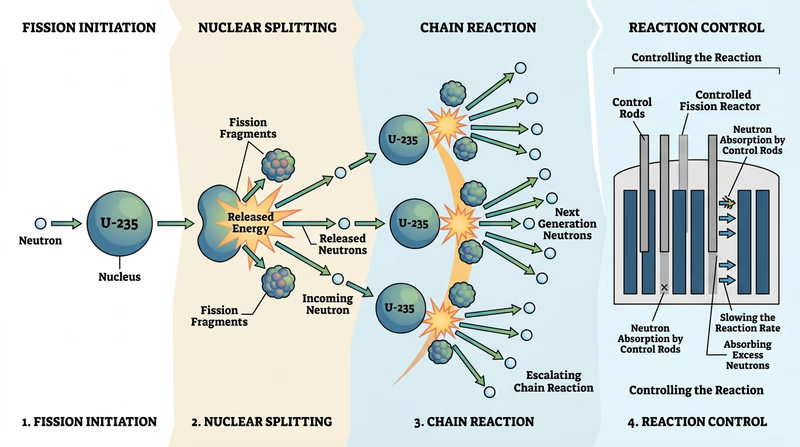
Req 6a — Fission and Chain Reactions
Nuclear fission happens when a heavy nucleus, such as uranium-235, absorbs a neutron and becomes unstable enough to split. When it splits, it releases energy, smaller nuclei, and more neutrons. Those neutrons can trigger additional fissions, which is how a chain reaction begins.
What your drawing should show
A clear fission drawing usually includes:
- the original heavy nucleus
- an incoming neutron
- the split into smaller nuclei
- released energy
- extra neutrons that can continue the process
How the mousetrap reactor helps
The mousetrap model is useful because it shows how stored energy can stay quiet until one event triggers many more. One launched ping-pong ball sets off multiple traps, which launch more balls, and the reaction grows quickly. It is not a perfect model, but it is a vivid one.
How a chain reaction is controlled
A reactor is designed so the chain reaction stays controlled instead of racing out of control. Engineers use control rods, moderators, coolant systems, and careful fuel arrangement to manage how many neutrons keep causing new fissions.
What critical mass means
Critical mass means enough fissile material is arranged so that, on average, each fission leads to one more fission and the chain reaction can continue. Too little material, or the wrong geometry, and too many neutrons escape.
🎬 Video: What Really Happened the First Time We Split a Heavy Atom in Half (video) — https://youtu.be/O2pyRDQgVjk?si=xYh6mItbfRQoaWqN
🎬 Video: What is Critical Mass | Nuclear Chain Reaction| Fission Chain Reaction| Critical Mass Of Uranium (video) — https://youtu.be/czjAtx8GjjA?si=l0xUwP2x5pJyQB85
🎬 Video: Mousetrap Fission (video) — https://youtu.be/vjqIJW_Qr3c?si=VFFlUgoPcuDMwqWh
The four ideas to explain clearly
These are the core teaching points of this requirement- Fission releases energy and neutrons
- Neutrons can trigger more fissions
- A reactor must control the reaction rate
- Critical mass depends on having enough fuel in the right arrangement
If you want to move from the physics of fission to the big system that powers homes and cities, the plant-focused option is next.
Req 6b — How Plants Make Electricity
This option takes you from nuclear physics to a full working energy system. A reactor is only one part of the story. To explain how electricity is made, you have to connect heat, steam, turbines, generators, transmission, and the energy mix around them.
How a plant makes electricity
In simple terms, a nuclear power plant uses fission to make heat. That heat turns water into steam or transfers heat to a steam system. The steam spins a turbine. The turbine turns a generator. The generator sends electricity to the grid.
That means nuclear plants are not magic machines that produce electricity directly from atoms. They are heat engines, just like many other power plants, but the heat source is a nuclear reaction instead of burning fossil fuel.
🎬 Video: What Is Nuclear Energy? Shrink Down to an Atom and Find Out (video) — https://youtu.be/6K5gy3RLcKc?si=sJl0OAEvTIGX7Pq-
What to find out during your visit or research
Questions your visit should answer
Use these to organize your notes- What kind of reactor or facility is it?
- How does heat move from the reactor to the turbine system?
- What safety and control systems are most important?
- How much electricity comes from nuclear power in the United States and in your state?
- What other major sources make up the rest of the energy mix?
Making the graph
Your graph should help your counselor compare sources quickly. A bar graph is often the easiest choice if you are comparing nuclear, natural gas, coal, hydro, wind, solar, and other sources between your state and the nation.
If your state has little or no nuclear generation, that is still worth discussing. It gives you a chance to explain how geography, policy, existing infrastructure, and local resources affect the energy mix.
The next requirement broadens the picture beyond power plants and shows how nuclear science helps people in many fields.
Req 7 — Nuclear Science in Daily Life
One of the best parts of this badge is realizing how often nuclear science helps people without drawing attention to itself. The field is not only reactors and warning symbols. It also helps doctors diagnose disease, engineers inspect materials, scientists track pollution, and spacecraft reach places where solar power is not enough.
Nuclear medicine
In nuclear medicine, doctors use radioactive tracers that collect in certain organs or tissues so cameras can show how the body is functioning. An example is a scan that helps doctors check blood flow in the heart or the function of the thyroid.
Environmental applications
Scientists can use isotopes to trace where water comes from, track pollution movement, study soil erosion, or learn how nutrients move through ecosystems. Nuclear techniques can reveal patterns that are hard to measure any other way.
Industrial applications
Industry uses radiation to inspect welds, test materials, measure thickness, sterilize equipment, and check whether sealed systems have leaks. One major strength is that radiation can help inspect the inside of something without cutting it open.
Space exploration
Some spacecraft use radioisotope power systems that produce electricity from the heat released by radioactive decay. These systems are valuable when a mission goes too far from the sun for solar panels to work well.
Radiation therapy
Radiation therapy uses carefully planned beams or radioactive sources to damage cancer cells while limiting harm to healthy tissue. This is one of the clearest examples of using ionizing radiation for a major human benefit.
🎬 Video: Radiation Technologies in Daily Life (video) — https://youtu.be/ePiNdzWjoWM?si=-VppE0yfL7AjUIN_
🎬 Video: Ways Nuclear Science Helps Cancer Patients (video) — https://youtu.be/zEihqK-jhBM?si=uLt8bBAD2BtJPBKJ
🎬 Video: Ways Nuclear Techniques Combat Malnutrition (video) — https://youtu.be/Wya79Ly8ec4?si=tCRoh8i2hF04R_F3
🎬 Video: Ways Nuclear Science Helps Ocean Health (video) — https://youtu.be/WYxo_iuzGYk?si=62AsgY7kDBUzLiZd
How to answer this requirement well
Use the same pattern for each application- Name a specific example
- Explain how nuclear science is used
- Say why the application matters to people, science, or society
If these applications sound interesting, the next requirement helps you explore the people behind them and the careers they build.
Req 8 — Explore a Nuclear Science Career
This requirement is your chance to connect the badge to a real person doing real work. Nuclear science careers are broader than many Scouts expect. Some professionals work at reactors or national labs, but others work in hospitals, universities, Navy programs, manufacturing, environmental monitoring, or radiation safety.
Good career areas to explore
Here are a few directions you could take:
- nuclear engineer
- health physicist or radiation safety officer
- medical physicist
- nuclear medicine technologist
- reactor operator
- research scientist
- radiochemist
- materials engineer or non-destructive testing specialist
What to learn about one career
Career research checklist
Bring these details to your counselor discussion- Training and education needed
- What the job costs to prepare for such as tuition, certification, or training time
- Job duties on a normal day
- Salary and job outlook
- How someone can advance in the field over time
- What about the career interests you or does not interest you
A good discussion is personal as well as factual. It is not enough to say a career pays well or needs a degree. Explain whether you would enjoy the kind of problems that person solves, the settings they work in, and the level of precision or responsibility the job requires.
🎬 Video: What do Nuclear Scientists Do? (video) — https://youtu.be/yedU3hs2cy8?si=_Dn1dycqxCP_X0EB
🎬 Video: Nuclear Engineering (video) — https://youtu.be/T5ptjDBGcP0?si=EJs0knZ2loLDV64Y
🎬 Video: Nuclear Medicine (video) — https://youtu.be/XcaChXkQmbM?si=Q4pbFu6uCxbkM2B_
🎬 Video: Navy Nuclear (video) — https://youtu.be/kU4Jzcj3bhM?si=4auD1J2itDYR8_8B
You have finished the badge requirements. The next page looks beyond them and shows where your curiosity can go next.
Extended Learning
Congratulations
You just worked through a badge that asks you to think at several scales at once: tiny particles, human health, giant machines, and society-wide energy choices. That is part of what makes nuclear science so interesting. The same ideas you used to explain an isotope can also help explain a cancer treatment or a power plant.
Why Background Radiation Matters
One of the best next steps is learning how scientists measure everyday background radiation carefully instead of speaking about it in vague terms. Where does it come from in your area? Does altitude matter? What role does radon play indoors? The more specific your questions become, the less mysterious radiation feels.
How Scientists Build Trust in Measurements
Nuclear science depends on confidence in measurements. Scientists have to calibrate instruments, understand uncertainty, compare readings, and avoid fooling themselves with bad setup or bad assumptions. If you liked the measurement parts of this badge, read more about detectors, calibration, and how evidence is tested before it is trusted.
Nuclear Science and Public Decisions
Few science topics connect to public policy as often as nuclear science. Communities debate medical access, plant licensing, waste storage, national security, and energy planning. A useful next step is learning how to separate evidence-based concerns from exaggeration, because responsible public decisions depend on that skill.
Real-World Experiences
Visit a Science Museum or Reactor Exhibit
Look for exhibits on atomic structure, radioactivity, power generation, or particle physics. A good exhibit can help you connect historical experiments to modern technology.
Tour a University Physics Department
Many universities host outreach events, engineering open houses, or public lectures. Even without a reactor on site, a physics department can show you how people enter the field.
Attend an Energy or STEM Career Event
Career fairs, utility open houses, and STEM expos can help you compare nuclear science with related fields such as chemistry, engineering, or environmental science.
Organizations
American Nuclear Society
American Nuclear Society Professional society resources, news, and career information for nuclear science and engineering. Link: American Nuclear Society — https://www.ans.org/International Atomic Energy Agency
International Atomic Energy Agency Global resources on peaceful nuclear applications, safety, medicine, agriculture, and energy. Link: International Atomic Energy Agency — https://www.iaea.org/U.S. Nuclear Regulatory Commission
U.S. Nuclear Regulatory Commission Public information on regulation, licensing, oversight, and nuclear safety in the United States. Link: U.S. Nuclear Regulatory Commission — https://www.nrc.gov/U.S. Department of Energy Office of Nuclear Energy
U.S. Department of Energy Office of Nuclear Energy Information on reactor technology, fuel cycle topics, innovation, and nuclear energy careers. Link: U.S. Department of Energy Office of Nuclear Energy — https://www.energy.gov/neNow that you have finished the full guide, the printable companion is ready when you want one page you can review offline or bring to a meeting.