Req 2 — Atoms, Isotopes, and Particles
This requirement gives you the vocabulary and models that make the rest of the badge easier to understand. First you learn the parts of matter and the language scientists use. Then you turn those ideas into something you can build and explain with your own hands.
Requirement 2a
A strong explanation connects the terms instead of listing them like flash cards.
An atom is the basic unit of an element. At its center is the nucleus, made of protons and neutrons. Electrons move around the nucleus in the surrounding electron cloud. Protons and neutrons are built from smaller particles called quarks.
An isotope is a version of the same element that has the same number of protons but a different number of neutrons. Some isotopes are stable. Some are unstable and change over time.
When an unstable nucleus changes and gives off energy or particles, that is radioactivity. A radioactive isotope is a radioisotope.
The main emissions you need to know are:
| Term | What it is | What to remember |
|---|---|---|
| Alpha particle | 2 protons + 2 neutrons | heavy, charged, stopped easily |
| Beta particle | fast electron or positron | lighter, travels farther than alpha |
| Gamma ray | high-energy electromagnetic radiation | no mass, very penetrating |
| X-ray | high-energy electromagnetic radiation usually produced outside the nucleus | also penetrating, often made by equipment |
Ionization happens when enough energy knocks electrons off atoms. Stability describes whether a nucleus tends to stay the same or decay.
Glossary of Nuclear Science Terms (website) A plain-language glossary you can use to check your definitions and make sure you are using each term correctly. Link: Glossary of Nuclear Science Terms (website) — https://nplab.webspace.durham.ac.uk/glossary/🎬 Video: What Are Isotopes? (video) — https://youtu.be/faB9Gb7bl9I?si=Et7Yi1cp8zLvyppP
🎬 Video: Quarks Explained (video) — https://youtu.be/LraNu_78sCw?si=o4zrlNUwhUT69eB6
Requirement 2b
This is where the badge stops being only words and turns into a model you can point to. Pick an element with several common isotopes so the pattern is easy to see. Hydrogen, carbon, oxygen, and chlorine all work well.
How to choose your element
Look for an element where the atomic number stays the same but the mass number can change. The atomic number tells you how many protons the element always has. The mass number is protons plus neutrons. Change the neutrons, and you have a new isotope of the same element.
What your three models should show
What to label on each isotope model
Make the differences easy to see
- Protons: these identify the element.
- Neutrons: these are what make one isotope different from another.
- Electrons: for a neutral atom, these match the proton count.
- Atomic number: the proton count.
- Mass number: protons plus neutrons.
- Isotope notation: for example, carbon-12 or (^12_6\text{C}).
A good set of models does not need to be fancy. Colored clay, beads, paper circles, or a digital drawing can all work if the structure is clear.
Showing quarks
In your separate model or diagram, show that:
- a proton is made of two up quarks and one down quark
- a neutron is made of one up quark and two down quarks
You do not need to explain every detail of particle physics. The goal is to show that protons and neutrons are not indivisible. They are made from smaller parts.
Make a Model of an Isotope (PDF) A step-by-step activity sheet that can help you organize your isotope models and notation. Link: Make a Model of an Isotope (PDF) — https://filestore.scouting.org/filestore/Merit_Badge_ReqandRes/Requirement%20Resources/Nuclear%20Science/Isotope%20Model%20Activity.pdf Interactive Isotopes (website) Lets you explore real isotope data and compare proton, neutron, and mass-number patterns before you build your model. Link: Interactive Isotopes (website) — https://isotopes.ans.org/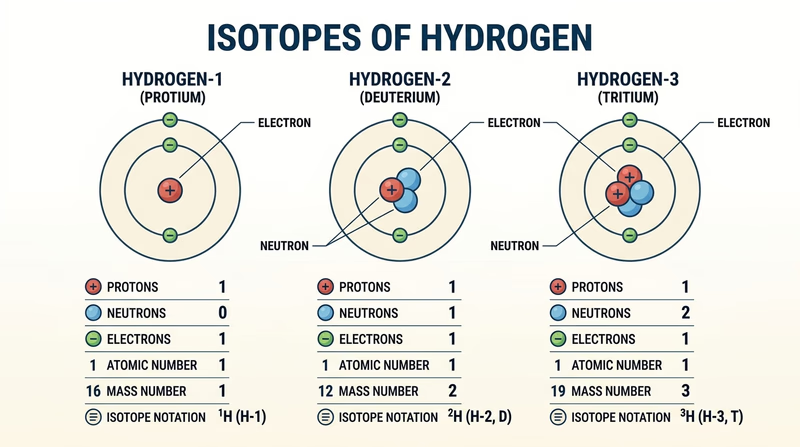
Once you are comfortable with atoms and isotopes, you are ready to look at machines that hurl particles at incredible speeds to learn even more.